We have explored up and down looking spectrometers in prior posts. In all cases the up looking instruments have been on the ground. A fun thing about the Modtran program is that you can look both up and down from any altitude you choose. Here we will begin at the surface and proceed up.
 Here we are looking at both up and down from 1 meter. The Modtran convention is that the “background run” is red and the “model” blue. In this series red will be looking down and seeing the radiation going up, and the blue will be looking up and seeing the radiation coming down.
Here we are looking at both up and down from 1 meter. The Modtran convention is that the “background run” is red and the “model” blue. In this series red will be looking down and seeing the radiation going up, and the blue will be looking up and seeing the radiation coming down.
It is interesting that looking down Modtran sees no signature of the CO2 bands, only a slight reduction in intensity and temperature from the surface blackbody over the range between WN’s 400-900. Looking up, there is a very strong signal from all the CO2 bands.
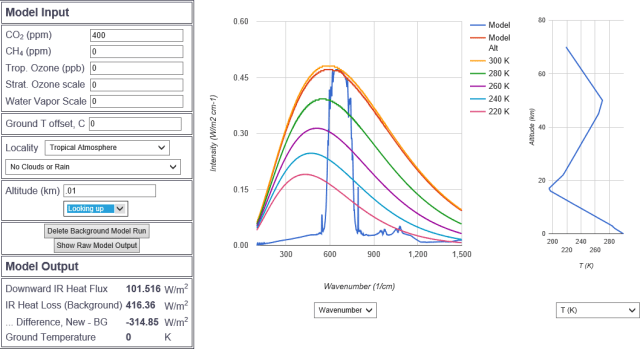
At ten meters there is very little difference.
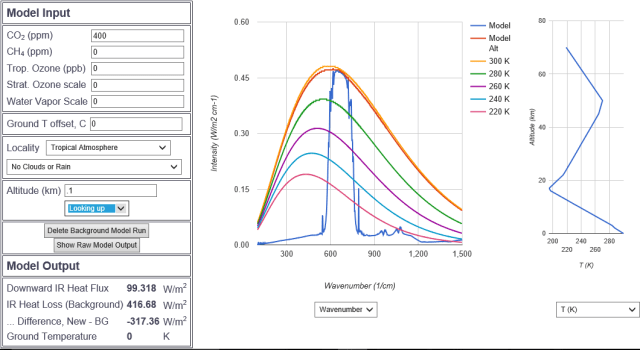
Deja vu all over again at 100 meters except for the suggestion of a signal at WN 618 where CO2 picks up a vibrational quantum number. This is the strongest CO2 band besides 667.4, albeit two orders of magnitude weaker.
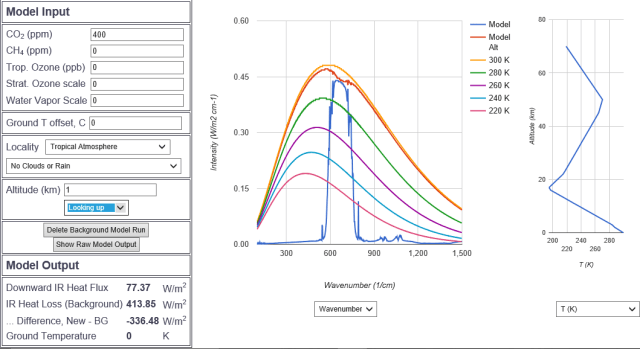
Finally, at one kilometer we begin to see a half baked signal in the CO2 bands looking down. How is this possible? How, in the first kilometer, where nearly all CO2 is introduced to the atmosphere, can there be no radiative signature?
The answer is simple and surprising. The atmosphere does not radiate in the CO2 bands in the first kilometer. At least not in the bands strong enough to generate a signal. In the first kilometer, all of the signal originates at higher altitudes.
CO2 is a fun kind of molecule. It likes to swim, and it likes to dance. Quantum vibration is the dominant form of excitation in the IR part of the spectrum and this “gitt’n jiggy wid it” is the reason CO2 absorption is thermalized rather that re radiated in the first kilometer of the earth’s atmosphere.
The concept here is very simple. We are talking about the percentage of light energy absorbed by CO2 that is kinetically dispersed (thermalized) rather than re emitted as a photon, and the observation that we are not seeing CO2 photons emitted in the first kilometer of the atmosphere.
The formal physics of this concept is very difficult. It engages the entire legal cannon of radiative transfer laws from Planck, Boltzmann, Kirchoff, Beer, et.al, without being satisfyingly resolved by any of them. An important reason for this is that the atmosphere is never in equilibrium. This concept of a tendency for CO2 to thermalize rather than radiate in the first kilometer is probably better thought of as just a physical property of the atmosphere.
We will climb higher in the next post.






Pingback: Modtran Up and Down II | geosciencebigpicture
Pingback: MODTRAN Up and Down IV, Upwards | geosciencebigpicture