There is a lot of confusion about emissivity. Emissivity is the tendency to emit; particularly the tendency to emit light after absorbing it. In the infrared part of the spectrum, where the earth emits radiation towards the atmosphere, there is very little scattering. Incident light is either absorbed, or it passes through.
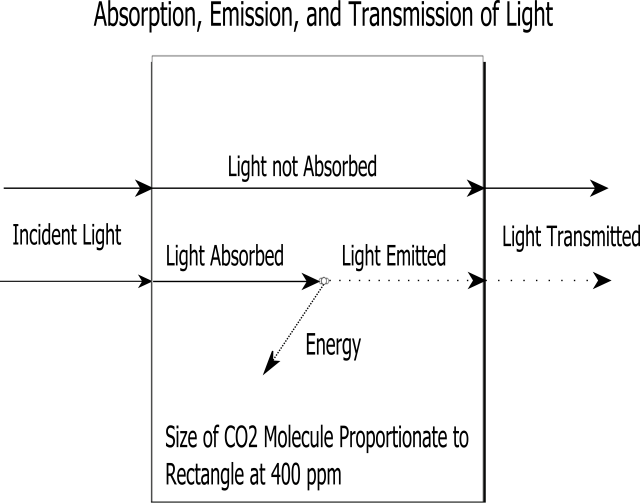
It can be seen that the light transmitted is equal to the light that passes unhindered plus the light that is emitted after absorption. The rectangle above can be thought of as a slab of atmosphere. Transmission is the incident light minus the transmitted light. Transmissivity is the transmitted light divided by the incident light. Emissivity is the proportion of light emitted to the light absorbed. For a perfect blackbody, emissivity is 1.
CO2 is far from a perfect blackbody.
Hottel and Leckner measured the column emissivity of CO2 in the 1940’s and 1970’s, respectively, at .14. Nasif Nahle has calculated a much smaller emissivity, .002, using two different methods. Staley and Jurica (1972) get .19. The range seems to be between poor and spectacularly poor emissivity, .002 to .2.
It turns out that in either case, the extremely high (98% in one meter) absorption of CO2 at 15 microns/wave number 667.4, and the poor transmission (2%), renders emissivity unimportant as regards upward radiation. The range of emissivity simply bounds the amount of light that passes straight through without being absorbed.

Above is the one meter absorption of CO2 at 400 ppm. The Q branch at 667 is 98% absorbed in one meter. This means that only 2% is transmitted. This transmission includes both absorbed and re-emitted light and unhindered light passing straight through.
Where emissivity becomes important is back radiation, as an equal amount to what is absorbed and re-radiated up, must be re-radiated downwards as well. The highest published full column emissivity is ~20%. This would seem to be the high limit of full column back radiation.
What about individual layers?
Staley and Jurica give a value of .08 for CO2 emissivity of a slab of one centimeter optical depth, a value of .14 for 10 centimeters, and a value of .19 for a meter. Optical depth is defined in several different ways by astronomers, chemists, and atmospheric scientists. Astronomers treat optical depth as the mean free path through a slab. In this treatment “mean free path”, “path length”, the distance a photon travels after entering before interaction, the average distance between interactions, the distance between the final interaction and escape, the distance you can “see” into the material, and optical depth; are essentially the same.
Optical depth is also defined as the path length times the absorption coefficient. The one meter absorption coefficient for the CO2 fundamental bend is .98. Path length is defined as partial pressure times the layer thickness. If the layer thickness is one meter, and the partial pressure is .04, the path length also becomes .04. We multiply this by the absorption coefficient to get an optical depth .04*.98*100(centimeters of CoE to a meter)=3.9
Beer’s Law defines optical depth as the negative natural log of transmittance. This is one of those mysterious empirical fits that work with surprising frequency. Transmittance is one minus whatever doesn’t get through (absorptance), so the negative natural log for a meter layer thickness and therefore the optical depth is -Ln(.02)=3.9. What luck.
The difference between the astronomical approach to optical depth and the chemistry and physics approach is that for astronomers optical depth is an actual distance, while the physical/chemical optical depth is dimensionless.
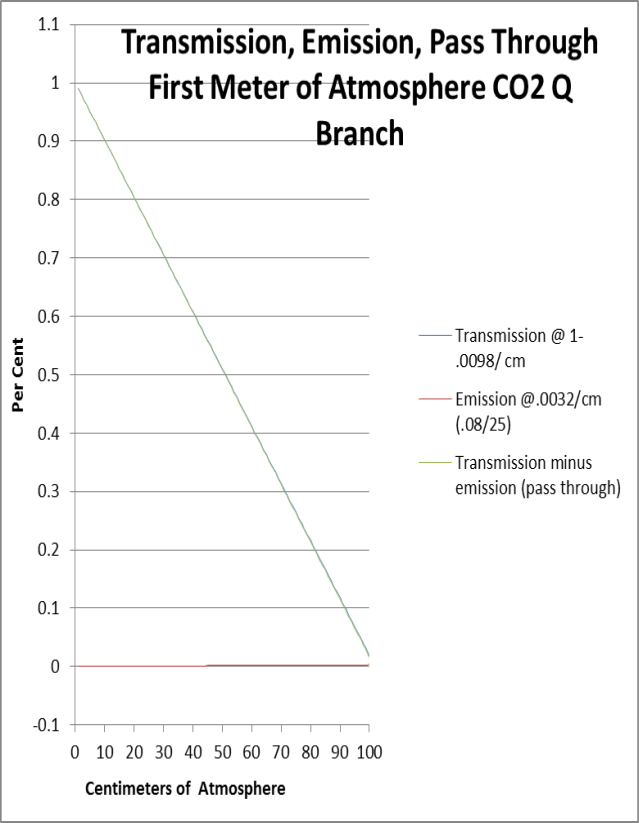
We can do a sanity check above where we plot linear values through a one meter slab by projecting centimeter scale values from measurements and calculations (.02 transmission, .08 emissivity/cm optical depth) We know perfectly well that none of this is linear, but transmission, and transmission minus emission are virtually indistinguishable. What this crude exercise can tell us is that it is very unlikely that any 667.4 photons pass through a one meter slab of atmosphere at current CO2 concentration. What we see transmitted through the slab is absorbed and re-emitted.
We can therefore assume that transmission, emission, and back radiation are all equal.
Back radiation must run the gauntlet of CO2 molecules on its way down as well. What we see radiated back down has the same mean free path to escape as what is transmitted up.
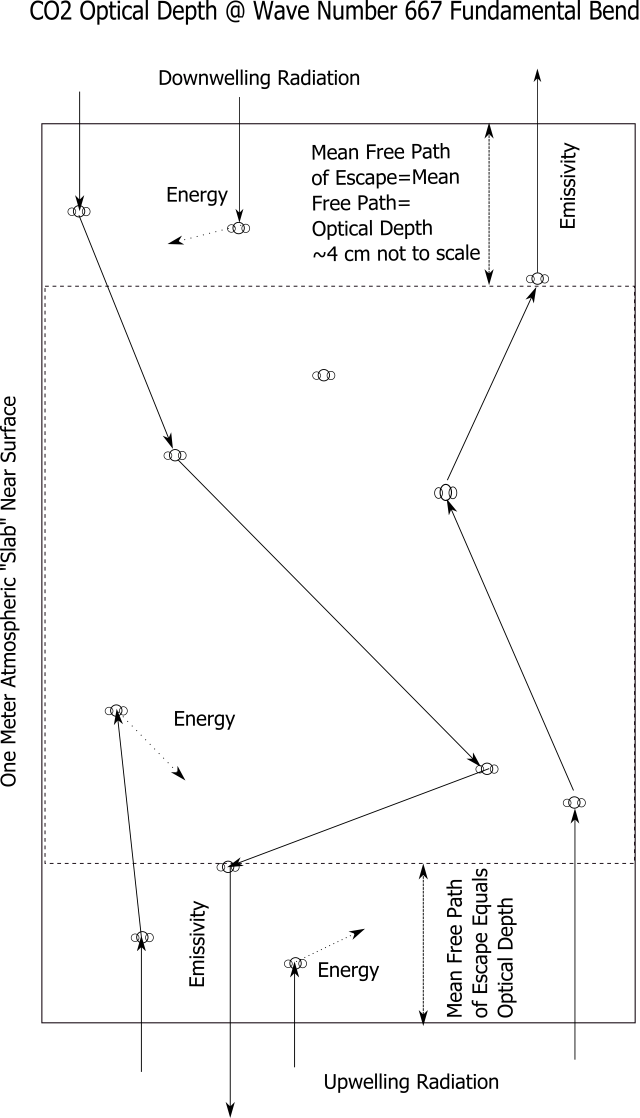
While the optical depth and free path remains the same up and down, the number of photons absorbed coming up, compared with those re-emitted in either direction, is reduced by the factor of emissivity. Back (downwelling) radiation can be no more than 2% of upwelling radiation.
The Schwarzschild equation used in radiative transfer models gets this completely backwards. The logic of this equation includes a “sink function” for absorption and a “source function” for re-radiation. This “source function” is given as the absorption coefficient, .98 in this case.
This would be true according to Kirchhoff’s Law if CO2 was a good blackbody with an emissivity of 1. We have seen that CO2 is a lousy blackbody with full column emissivity somewhere between .002 and .2. The value we have developed above, .02, falls in this range.
The “source function” in the Schwarzschild equation must be very significantly adjusted downward to reflect the real world emissivity of CO2.






Peter Grimshaw commented on Emissivity of CO2
There is a lot of confusion about emissivity. Emissivity is the tendency to emit; particularly the tendency to emit light after …
Hi!
I’ve written a piece using Dalton partial pressure laws and how partial pressures relate to radiation. I am trying to make sense of radiative issues, transmission, internally generated IR and so on.
I wonder if you have time to take a look?
My main thesis is that a greater density of CO2 molecules in the atmosphere must generate MORE IR quantum events.
So More CO2 means More IR, More IR means more IR is lost to space at Top of Atmosphere, so this means More COOLING at top of atmosphere.
Obviously this is in direct contradiction with AGW theory.
Or am I missing something really simple and basic, ie the Emissivity of CO2 in the atmosphere taken as a whole is ‘1’ which I think it is, and that somehow affects my modelling.
Anyway, would be great if you could take a look.
Your article above seems very grounded.
Thankyou in any case!
Peterg Uk
Here is my article :-
More CO2 means More IR, More Cooling?
Does Dalton’s Law turn the Greenhouse Gas effect on it’s head?
Is Carbon Dioxide an atmospheric COOLANT?
The general view of climate scientists is that more CO2 WARMS the atmosphere.
But does Dalton suggest more CO2 leads to MORE IR leaving the atmosphere ?
More CO2 molecules means more radiative events?
The key mechanism Anthropocentric Global Warming (AGW) thinkers describe is to use is something called EEH, Effective Emission Height.
The EEH is a real phenomenon.
It is caused in part by the CO2 gas being denser
It is fair to say EEH IS generally believed to be the key ‘Global Warming’ mechanism.
And it works as AGW proponents say it does.
It uses the decrease in temperature as you go higher in the atmosphere to reduce radiation.
It can be subtle and tricky to compute or explain (http://clivebest.com/blog/?p=4597) but it works, and it would REDUCE IR emissions from CO2 leaving the atmosphere, IF a fixed amount of radiation was flowing through the atmosphere.
Essentially, the upward IR comes from a colder place, so less IR.
If it was a fire, we would say the fire had grown a bit colder.
But there are three mistakes it seems this argument is making –
1. Trying to explain atmospheric heat transfer using radiation.
2. Thinking of radiation as a fixed quantity of energy being transported.
3. Not realising the IR implications of CO2 being a trace gas increasing in density.
As CO2 levels in the atmosphere increase, the internal generated IR also rises simply due to the fact there is a denser concentration of molecules. This means denser IR.
This is the bit that is missed, probably because people are focussed on the transmission of a set amount of energy ‘through’ the CO2.
If this was a fire, we could almost say the fire had got bigger, there is more of it.
So the point is radiation is partly a factor of the density of gas. Emissivity increases with density.
Double the CO2 in the atmosphere, and pretty much, you double the radiative output, and this is NOT factored in to most AGW calculations, (including Clive – see link above – clever Clive also forgets this because he is working on an absorption model).
Dalton’s law implies that double the molecules of CO2 will double it’s partial pressure (https://en.wikipedia.org/wiki/Dalton%27s_law).
And increasing partial pressure increases radiation.
See the table at the bottom of this article.
A word on radiation.
The radiation emitted by the atmosphere is completely independent of the amount of radiation it is receiving. The amount of “upwelling IR is of no consequence when calculating how much IR the gas will emit.
Radiation from a gas is described by the SB (Stephan-Boltzman) rules and is dependent on the temperature of the gas and the quantity (or ’emissivity’), NOT the amount of radiation upwelled or that it receives. Thinking about radiative ‘heat transfer’ confuses the issue, so let’s stop.
So, in conclusion, increasing CO2 in the atmosphere has two effects.
They are both to do with what might be called a ‘grey’ body becoming a bit more ‘black’.
The trace gas becomes a bit more denser, more opaque, a bit more solid.
And this results in two effects –
1 An increase in EEH, which is real and lowers outgoing IR a bit, the fire cools, a bit.
2. An increase in IR from an increased amount of CO2. The IR fire almost doubles.
So the IR fire cools, but gets lots bigger.
I also think the key heat transport mechanism in the atmosphere is probably not radiation, it is convection at the equator, where the sun is hottest. So we need to let go of the idea of any heat being ‘blocked’. Rising air transports massive amounts of energy from the bottom to top of atmosphere where it is stored as potential energy. This creates atmospheric pressure, and is slowly released to outer space by radiation. And this radiation is simply a property of the gas at TOA. It does not mean the energy has been radiated “through” the atmosphere. Think tropical thunder storms for the power of convective heat transport. And compare with cold ice caps where there is little sun. Radiation doesn’t create thunder storms, sun + convection does, and this transports loads of energy to TOA where it is stored as potential energy. If someone knows how much that would be good? I appreciate this paragraph is woolly.
The met office even has a name for this heat-storage mechanism is CAPE – Convective Available Potential Energy https://en.wikipedia.org/wiki/Convective_available_potential_energy
Thinking in terms of radiation transporting heat and being ‘blocked’ does not well describe how our atmosphere works and confuses thinking. Is convection the main heat transport mechanism?
CO2 seems to be, believe it or not, is only ever a coolant.
Here is a table which describes increasing CO2 emissivity with increase pressure/density
More CO2 means More IR will leave our atmosphere.
These figures were calculated about 60 years ago by a man called Hottel.
http://www.biocab.org/Emissivity_CO2.html
More partial pressure = More radiation.
More CO2 cools the air even more.
Peter Grimshaw
info@clearaccountingadvice.co.uk
Approve Trash | Mark as Spam
Thanks Peter. (I posted your comment myself after WordPress declined) I agree, but would like to see a calculation of the magnitude of your effect. The kinetic transfer of energy is also neglected in the radiation only approach. Kinetic transfer dominates the first half kilometer of the atmosphere which radiates like a brick at the Planck curve.
I want to recommend a recent paper using CERES data. A surprising observation is that the GHE only works as advertised in clear sky areas which typically cover only 1/3 of the earth’s surface. More surprisingly, GHE breaks down in cloudy areas and radiative loss to space is increasing above them. Perhaps your effect?
https://www.mdpi.com/2073-4433/12/10/1297
I find no numbers for emissivity of gases. Since our native gases are colorless, they should have emissivity near zero. So CO2 should have no more impact on global warming than any other. Global warming comes from the heat of formation of fossil fuels that can’t be readily sequestered as does the heat of formation of biofuels.
Everything with a temperature above absolute zero emits light, almost always in the same bands it absorbs. Different materials have different emissive properties. Some like to spit more photons after absorbing radiative energy. Some prefer to dance. CO2 prefers to dance. Color is irrelevant. Only a very few can see IR.