Carbon Isotope Excursions and Carbon Limitation of Primary Productivity in the Biosphere
Gordon Lehman, Trunkmonkey, Gymnosperm; sheepherders, ignoramuses all
Abstract
The conception of natural history as an economic struggle was developed by Geerat Vermeij (1). “Supply side” interpretations of δ13C excursions in the carbonate shells of marine organisms have been forced to invoke methane hydrates, exponential increases in bacterial carbon remineralization, improbable rates of organic carbon transfer from land, and even carbon laden comets to account for a superabundance of the lighter δ12C preferred by carbonate producing organisms during negative δ13C events, and a superabundance of δ13C during positive events. We propose here a “demand side” approach to δ13C excursions supposing that primary productivity in the biosphere is carbon limited.
Introduction
While volcanism produces infinitesimal amounts of free oxygen and dissociation of water by ultraviolet light a bit more, it is widely accepted that the preponderance of the oxygen, comprising nearly 21% of the atmosphere, was produced by photosynthesis. The equation for photosynthesis is 6CO2 +6 H2O -> C6H12O6 + 6O2. Every pair of oxygen atoms in the atmosphere consumed a molecule of CO2 at its formation.
Photosynthesis has been around more than three billion years but oxygen levels in the atmosphere are thought to have remained below 1% until the banded iron formation/red bed (magnetite/hematite) transition about two billion years ago when oxygen concentration in the atmosphere may have risen to 10%. The BIF/red bed transition corresponds generally with the evolution and endosymbiotic assimilation of mitochondria, which used oxygen to first double the ATP efficiency of cellular respiration through the Krebs cycle and eventually turbocharge respiration by an order of magnitude with oxidative phosphorylation.
The formula for mitochondrial respiration is C6H12O6+ 6 O2 → 6 CO2+ 6 H2O. Being equal and opposite to photosynthesis, a sustainable economy had evolved which allowed life to proliferate for nearly two billion years and eventually colonize the land.
About 375 million years ago in the late Devonian substantial forests of tall trees covered land for the first time. We suppose that this event marks the beginning of a striking increase in O2 and depletion of CO2 that persisted until the end of the Paleozoic. We further suppose that until plants had substantially colonized land, volcanic carbon production (2) had been sufficient to balance the demand from oceanic photosynthesis and the other biological and chemical demands, but the added carbon demand from land plants exceeded the supply. Since the late Devonian carbon and oxygen in the atmosphere have been inversely correlated.
Figure 1 from Huey and Ward (6) using data from Robert Berner shows Phanerozoic O2 and CO2 levels expressed as percentage of current levels. Current levels are shown at the left and the vertical red line is the Permian-Triassic extinction.
We offer no explanation for the Permian-Triassic extinction, but suppose that reduced carbon demand and photosynthesis allowed biological respiration to predominate, depleting oxygen and restoring CO2. Late Permian strata contain anomalously high concentrations of fungal spores (7).
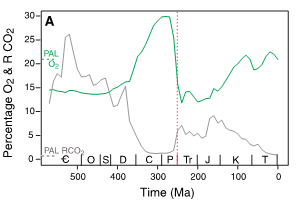
Figure 1 by Huey and Ward (6)
Large amounts of biological carbon are stored in the geosphere as biomass (predominantly cellulose and lignin); fulvic and humic acids and humins; as carbonate, CaCO4; as methane hydrates and as kerogens of various hydrocarbon compositions. It is estimated that the lithosphere contains 75 million gigatons of carbon as carbonate and kerogens (3). Inorganic carbon is stored as carbonic acid, H2CO3, and its alkaline chemical weathering products like evaporate basins, kaolin clays, and other weathered alkaline sedimentary rocks. All of these biological and chemical processes represent current and historic demands on the carbon supply.
While some free oxygen is consumed in chemical weathering, the vast majority of oxygen stored on carbonate and other geological carbon sinks was produced directly from water. Water vapor comprises 37% to 97% of the volume of volcanic emissions (4). It is generally accepted that the water on the planet is mineral and did not consume atmospheric free oxygen in its formation.
A small amount of free oxygen is converted to ozone (O3) by ultraviolet light, but ozone has a half-life of hours, it degrades back to free oxygen, and at .6 parts per million (5) in the atmosphere ozone is safely ignored for our purposes.
We suppose that oxygen has accumulated in the atmosphere because cellular respiration, the only significant demand on it, produces a biologically valuable end product (CO2) that is quickly recycled; and that carbon is depleted in the atmosphere because it faces many biological and chemical demands with longer cycle times. We find it significant that at .04% current CO2 concentration in the atmosphere is nearly four orders of magnitude lower than the concentration of free oxygen.
Carbon Isotope Excursions
Seasoned colleagues can remember when a university department was lucky to have a mass spectrometer. The proliferation of these instruments revolutionized science in the late twentieth century and geology benefitted as much as any field of inquiry. Measurements have been taken of the carbon isotope ratios in the carbonate shells of organisms and the ages of these organisms determined by the decay of other isotopes. Most of this work has focused on marine organisms, but is assumed that on geological timescales the ocean and atmosphere equilibrate. It was noticed that over time carbon isotope ratios were pretty constant but took some surprising excursions.
Work is mass over distance and weight is an economic cost. Natural selection has favored organisms that minimized costs, and biological processes select lighter isotopes when they are available. It has become standard practice to plot carbon isotope variation as deviation or anomaly of δ13C, the heavier isotope. Figure 2 by Jonathan Payne et al (8) shows the δ13C excursions of the late Permian and the Triassic. Lower numbers (movement to the left) in δ13C mean that less δ13C and more δ12C is incorporated in carbonate shells. Supply side interpretations of negative excursions postulate enormous influxes of δ12C to account for this shift. We suppose the shift reflects reduced competition for δ12C. It is notable that the negative excursion in the late Permian was only the beginning of a roughly six million year period in the early Triassic of even higher amplitude excursions. We interpret these swings as fluctuations in global carbon demand, with positive δ13C periods coinciding with biological recovery by colonizing (r selected) species and higher carbon demand, and negative δ13C periods coinciding with extinction and lower carbon demand. General recovery from the Permian-Triassic extinction did not begin until after δ13C had stabilized in the mid-Triassic.
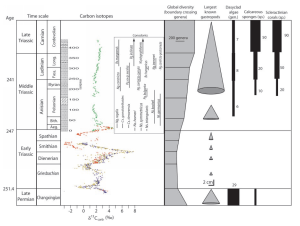
Figure 2 by Jonathan Payne et al (8)
Figure 3 by Saunders and Reichow (9) shows argon isotope timing of the negative δ13C excursion in relation to the extinction horizon. We interpret the coincidence of the negative excursion and the extinction to reflect the massive reduction in photosynthesizing and carbonate producing organisms and reduced demand for carbon in general and δ12C in particular. The remaining organisms would have had more δ12C available and would have included less δ13C in their shells.
Stephen Stanley (10) has found as many as 26 Phanerozoic extinctions associated with isotopic excursions. In all of these the concentration of the heavy oxygen isotope δ18O, similarly selected against in biological processes, covaries in the carbonate shells. We suppose that the isotope excursions did not cause the extinctions, but merely reflect them.
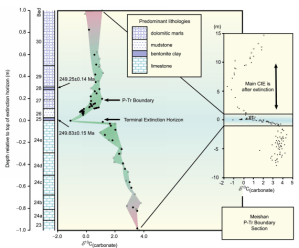
Figure 3 by Saunders and Reichow (9)
Figure 4 by Thomas and Shackleton (11) shows the oxygen and carbon excursions during the marine extinction near the much discussed Paleocene-Eocene thermal maximum (PETM). Please note that the δ18O scale is inverted and the δ13C and δ18O excursions have the same sign. This time series is especially interesting because it also spans the Cretaceous-Tertiary (K/T) extinction of the dinosaurs. One can see a modest signal at about 65 ma. We notice some similarity with the Permian-Triassic pattern where an initial excursion is followed by higher amplitude events before settling down.
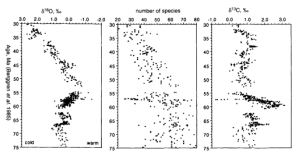
Figure 4 by Thomas and Shackleton (11)
Conclusion
We conclude that attempts to understand the relationship between isotope excursions and extinctions have been hampered by a preconception, tied to current notions of carbon and warming, that the excursions must have caused the extinctions. We suppose the opposite, that isotopic excursions reflect demand that waxes and wanes with the abundance of carbon limited organisms. Wolfe-Gladrow et al (12) and Reibesell et al (13) have reported striking increases in plankton productivity when carbon supply is increased. It is widely recognized that the carbon double bond is the chemical basis of life on our planet. We find it easy to suppose that primary productivity is carbon limited.
References
(1)Vermeij, G. (2006). Nature: an economic history. Princeton University Press. Princeton University Press. Retrieved from http://books.google.com/books?hl=en&lr=&id=H9RFCa7rH8QC&oi=fnd&pg=PR1&dq=Nature-+an+economic+history&ots=-E_eNzhLjW&sig=5yqmcTCu2iCSRpTkN6OiJgra9v0
(2) Symonds, R.B., Rose, w.I., Bluth, G., and Gerlach, T.M., 1994, Volcanic gas studies: methods, results, and applications, in Carroll, M.R., and Holloway, J.R., eds., Volatiles in Magmas: Mineralogical Society of America Reviews in Mineralogy, v. 30, p. 1-66.
(3) Lal, Rattan (2008). “Sequestration of atmospheric CO2 in global carbon pools”. Energy and Environmental Science 1: 86–100. DOI:10.1039/b809492f.
(4) Sigurdsson, H. et al., (2000) Encyclopedia of Volcanoes, San Diego, Academic Press
(5) “Science: Ozone Basics.”. http://www.ozonelayer.noaa.gov/science/basics.htm. Retrieved 2007-01-29.
(6) Huey, R. B., & Ward, P. D. (2005, April 15). Hypoxia, global warming, and terrestrial late Permian extinctions. Science (New York, N.Y.). doi:10.1126/science.1108019
(7) H Visscher, H Brinkhuis, D L Dilcher, W C Elsik, Y Eshet, C V Looy, M R Rampino, and A Traverse (1996). “The terminal Paleozoic fungal event: Evidence of terrestrial ecosystem destabilization and collapse”. Proceedings of the National Academy of Sciences 93 (5): 2155–2158. Bibcode 1996PNAS…93.2155V. DOI:10.1073/pnas.93.5.2155. PMC 39926. PMID 11607638. //www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=39926.
(8) Payne, J. L., Lehrmann, D. J., Wei, J., Orchard, M. J., Schrag, D. P., & Knoll, A. H. (2004). Large perturbations of the carbon cycle during recovery from the end-permian extinction. Science (New York, N.Y.), 305(5683), 506-9. doi:10.1126/science.1097023
(9) Saunders, A.D and Reichow, M.K. 2009. The Siberian Traps and the End-Permian mass extinction: A critical review. Chinese Science Bulletin, 54, 20-37. Download pdf.
(10) Stanley, S. M. (2010). Relation of Phanerozoic stable isotope excursions to climate, bacterial metabolism, and major extinctions. Proceedings of the National Academy of Sciences of the United States of America, 107(45), 19185-9. doi:10.1073/pnas.1012833107
(11) Thomas, E., & Shackleton, N. J. (1996). The Paleocene-Eocene benthic foraminiferal extinction and stable isotope anomalies. Geological Society, London, Special Publications, 101(1), 401-441. doi:10.1144/GSL.SP.1996.101.01.20
(12) Wolf-Gladrow, B. D. A., Riebesell, U. L. F., & Burkhardt, S. (1999). composition of marine plankton, 461-476.
(13) Riebesell, U., Wolf-Gladrow, D. a., & Smetacek, V. (1993, January 21). Carbon dioxide limitation of marine phytoplankton growth rates. Nature. doi:10.1038/361249a0
Permisions:
Figure 1
Figure 2
Licensee: Gordon
Lehman
License Date: Jul 27, 2012
License Number: 2956900028735
Publication: Chinese Science Bulletin
Title: The Siberian Traps and the End-Permian mass extinction: a critical
review
Figure 3
Figure 4
By policy of Geological Society, London.